People with too much body fat, either overweight or lean but metabolically obese (AKA skinny fat) are more susceptible to infections and related complications, periodontal disease, and even autoimmunity. Common inflammatory diseases linked with excess body fat include rheumatoid arthritis (RA), systemic lupus erythematosus (SLE), inflammatory bowel disease (IBD), multiple sclerosis (MS), type-1 diabetes (T1D), psoriasis and psoriatic arthritis (PsA), and auotimmune thyroid disease (i.e.Hashimoto thyroiditis). But why?
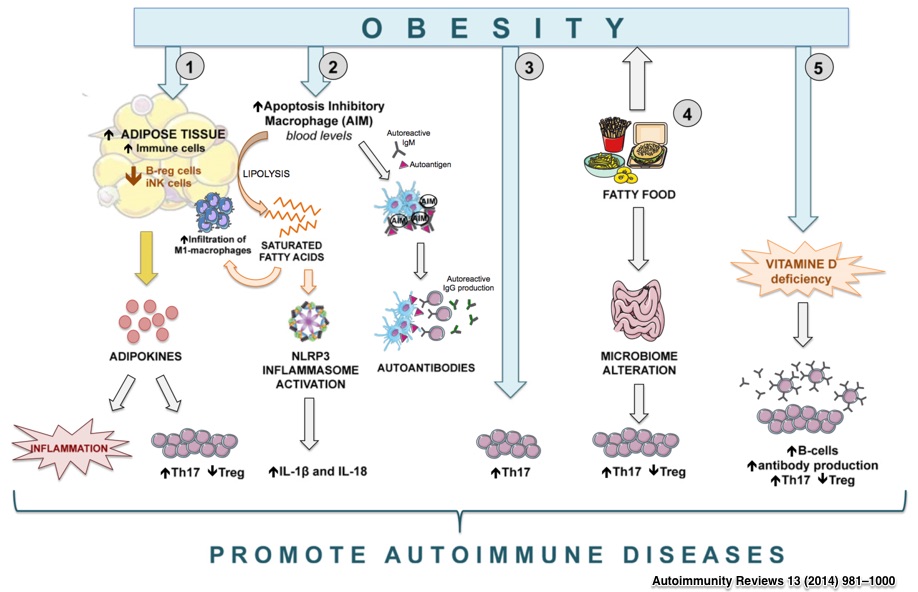
It turns out that belly fat, is sick. This deep, intra-abdominal or visceral adipose tissue (VAT) is very close to intestinal organs and is highly inflammatory. (In contrast, subcutaneous adipose tissue (SAT) on the upper arms, back, legs, and buttocks has little, if any, harmful immune or metabolic effects.)
Unlike subcutaneous fat of the upper arms, visceral fat tends to attract metabolically disruptive immune cells such as macrophages and lymphocytes. These immune cells release inflammatory messengers, or cytokines, that antagonize your metabolic machinery, disrupting your fat-loss efforts and creating a vicious cycle of more inflammation and fat accumulation. Unsurprisingly, an apple shape is associated with many of the metabolic and inflammatory diseases that occur with increased body fat percentage. This is why I call it “sick fat.”
Why is Belly Fat So Inflammatory
There are four main ways that fat increases inflammation:
- Increased leptin release (which impacts Treg cells)
- Increase adipose tissue hypoxia
- Reduced adiponectin release
Lets discuss in more depth.
Enlarged Fat Cells Become Devoid of Oxygen, Stimulating Inflammation
As fat cells enlarge when a person gains weight, progressing from lean to overweight to obese, oxygen availability to cells declines. This, in combination with other cellular stress messengers, triggers a key signaling molecule called hypoxia-inducible factor 1 alpha (HIF-1α). Increased HIF-1α is linked to changes in the inflammatory predisposition of immune cells, such as increased type 1 macrophage and the Th-17 type of T lymphocytes. This “immune signature” is linked to metabolic abnormalities, such as impaired fat burning and insulin resistance.
The combination of increased leptin and low oxygen levels in fat tissues that increase HIF-1α. That response, in combination with the surge of leptin, suppresses the immune cell guardians, the Treg cells that keep other T cells and inflammatory macrophages at bay.
By default, hypoxia is incongruent with oxygen-dependent fat burning, and so the only way immune cells can meet their increased energy demands in response to inflammation promoters like endotoxin, or HIF-1α from low oxygen, is to burn sugar as fuel.
Reduced Leptin Signaling Reduces Treg Cells Around Fat Tissue, Driving Inflammation
Yes. It’s complicated. Here’s another way to look at it: in their healthy state, working tissues, including immune cells, use oxygen to burn fat. For example, some immune cells (M2 macrophage, Th2, and Treg cells) rely heavily on fat for energy. Not so for the metabolically challenged, insulin-resistant, inflamed immune cells in fat tissue, the ones that are characteristic of obesity. These cells, which make up about 40 percent of fat tissue, shift out of fat-burning mode into sugar-burning mode.
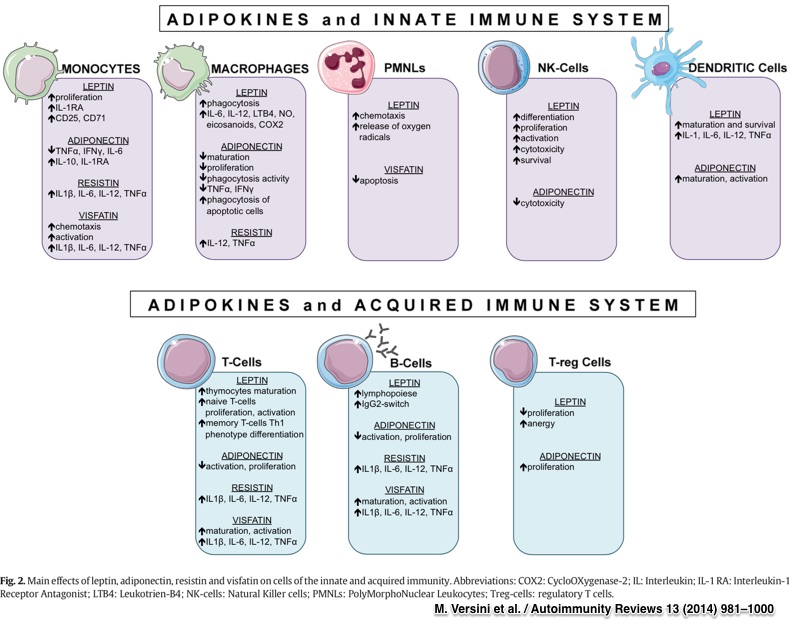
Inflammatory triggers such as bacterial endotoxin favor immune cells that thrive on sugar burning, including the M1-type macrophage and Th1 and Th17 types of T helper cells. Unfortunately, these cells also induce inflammation, which is counterproductive to insulin sensitivity and fat burning.
In contrast, our steady-state anti-inflammatory cells, including the M2 macrophage and Treg cells, are great fat burners. Their existence is heavily dependent on proper levels of the mitochondrial regulatory factors discussed above, including AMPK.
Summary and Conclusion
Belly fat is highly inflammatory and is linked with many inflammatory diseases. Worse yet, the immune signature of inflammation (from increase fat tissue) creates an situation where sugar burning is attenuated, in favor of fat burning; further creating inflammation.
Whether fat loss is a goal, or reducing inflammation and recovering from autoimmune disease is your goal, targeting both the fat cells and immune system are key tactics to reclaim balance in your body.
In my book Belly Fat Effect: The Real Secret About How Your Diet, Intestinal Health, and Gut Bacteria Help You Burn Fat I discuss many novel strategies to help accomplish both of these goals. You can preview the book right here on Amazon.com

http://www.burberry-factory.net/
http://www.shophandbagsonline.com/
http://www.official-coachoutlet.com/
http://www.barbour-factory.com/
http://www.burberry-outlet2014.com/
http://www.guccibags.us.com/
http://www.marcjacobsonsale.com/
http://www.mcmworldwide.ca/
http://www.guccishoes-uk.com/
http://www.kate-spades.com/
http://www.louisvuittonas.com/
http://www.lv-guccishoesfactory.com/
http://www.official-mkoutlets.com/
http://www.official-pradaoutlet.com/
http://www.michael-korsusa.net/
http://www.north-facesoutlet.com/
http://www.moncler-clearance.com/
http://www.north-faceclearance.com/
http://www.clothes-mall.com/
http://www.polo-outlets.com/
http://www.ralphlauren.so/
http://www.ralphlaurentshirts.com/
http://www.ferragamos.in.net/
http://www.longchampsoutlet.com/
http://www.abercrombiee.com/
http://www.barbour-jacketsoutlet.com/
http://www.michael–korsonline.com/
http://www.thenorthface.so/
http://www.beatsbydreoutlet.net/
http://www.tommyhilfiger.in.net/
http://www.ralphslauren.co.uk/
http://www.michaelkors.so/
http://www.oakleyssunglassoutlet.com/
http://www.warm-boots.com/
http://www.woolrich-clearance.com/
http://www.tommy-hilfigeroutlet.com/
http://www.official-northfaceoutlet.com/
http://www.nike-jordanshoes.com/
http://www.monsterbeatsbydres.net/
http://www.canada-gooser.com/
http://www.bestcustomsonline.com/
http://coach.mischristmas.com/
http://www.coach-blackfriday2014.com/
http://www.coachccoachoutlet.com/
http://www.coach-clearance.com/
http://www.coach-factories.net/
http://www.coach-factorysoutlet.com/
http://www.coachlosangeles.com/
http://www.coachoutletstates.com/
http://www.coach-pursesoutlets.com/
http://www.hermes-outletonline.com/
http://www.misblackfriday.com/
http://www.mischristmas.com/
http://www.mmoncler-outlet.com/
http://www.newoutletonlinemall.com/
http://www.ralphlaurenepolo.com/
http://www.zxcoachoutlet.com/
http://michaelkorsoutlet.mischristmas.com/
http://mcmbackpack.mischristmas.com/
http://monsterbeats.mischristmas.com/
http://northfaceoutlet.mischristmas.com/
http://mk.misblackfriday.com/
http://coachoutlet.misblackfriday.com/
http://coachfactory.misblackfriday.com/
http://uggaustralia.misblackfriday.com/
http://coachpurses.misblackfriday.com/
http://coachusa.misblackfriday.com/
http://coach.misblackfriday.com/
http://michaelkorss.misblackfriday.com/
http://michaelkors.misblackfriday.com/
http://airmax.misblackfriday.com/
http://michael-kors.misblackfriday.com/
https://twitter.com/CoachOutlet2014
https://www.facebook.com/coachoutletstoreonline
https://www.facebook.com/ralphlaurenoutletonline
My pal suggested I'd personally perhaps in this way web page. They once were completely right Rose Gold Wedding Rings. The following send truly made our day. You cannot think about precisely how much time frame I needed spent for this information! Thanks a lot!
Michael Kors UK Outlet – MK bags and purses cheap on sale online!
Now free global shipping for buying handbags in our Michael
Kors Outlet and save most up to 81% off!
more from : michael kors outlet uk
Hello Web Admin, I noticed that your On-Page SEO is is missing a few factors, for one you do not use all three H tags in your post, also I notice that you are not using bold or italics properly in your SEO optimization. On-Page SEO means more now than ever since the new Google update: Panda. No longer are backlinks and simply pinging or sending out a RSS feed the key to getting Google PageRank or Alexa Rankings, You now NEED On-Page SEO. So what is good On-Page SEO?First your keyword must appear in the title.Then it must appear in the URL.You have to optimize your keyword and make sure that it has a nice keyword density of 3-5% in your article with relevant LSI (Latent Semantic Indexing). Then you should spread all H1,H2,H3 tags in your article.Your Keyword should appear in your first paragraph and in the last sentence of the page. You should have relevant usage of Bold and italics of your keyword.There should be one internal link to a page on your blog and you should have one image with an alt tag that has your keyword….wait there's even more Now what if i told you there was a simple WordPress plugin that does all the On-Page SEO, and automatically for you? That's right AUTOMATICALLY, just watch this 4minute video for more information at. <a href="http://www.SEORankingLinks.com">Seo Plugin</a>
How Belly Fat Causes Inflammation and Promotes Autoimmune Diseases
[url=http://www.gapm9n4f6j61i7a644d1j75xu72zfy25s.org/]udfqtqcrt[/url]
dfqtqcrt http://www.gapm9n4f6j61i7a644d1j75xu72zfy25s.org/
<a href="http://www.gapm9n4f6j61i7a644d1j75xu72zfy25s.org/">adfqtqcrt</a>
How Belly Fat Causes Inflammation and Promotes Autoimmune Diseases
[url=http://www.gx1nl06t605g369m4bac4q2ds33lk43js.org/]uzfwygrov[/url]
<a href="http://www.gx1nl06t605g369m4bac4q2ds33lk43js.org/">azfwygrov</a>
zfwygrov http://www.gx1nl06t605g369m4bac4q2ds33lk43js.org/
granger whitelaw shares his thoughts on life, love, family and friends.
How Belly Fat Causes Inflammation and Promotes Autoimmune Diseases
<a href="http://www.ge7olaix7495it7idl2t55559dpn8651s.org/">atnhcjpjdgm</a>
tnhcjpjdgm http://www.ge7olaix7495it7idl2t55559dpn8651s.org/
[url=http://www.ge7olaix7495it7idl2t55559dpn8651s.org/]utnhcjpjdgm[/url]
How Belly Fat Causes Inflammation and Promotes Autoimmune Diseases
[url=http://www.g8xn6r20692rloez3c1m5g4u93nv41h9s.org/]uysvqfje[/url]
<a href="http://www.g8xn6r20692rloez3c1m5g4u93nv41h9s.org/">aysvqfje</a>
ysvqfje http://www.g8xn6r20692rloez3c1m5g4u93nv41h9s.org/
How Belly Fat Causes Inflammation and Promotes Autoimmune Diseases
nvcbcxziqw http://www.gnd2m6h69kj479m5hanbv5k8t3692t26s.org/
<a href="http://www.gnd2m6h69kj479m5hanbv5k8t3692t26s.org/">anvcbcxziqw</a>
[url=http://www.gnd2m6h69kj479m5hanbv5k8t3692t26s.org/]unvcbcxziqw[/url]
I am very new to web design as I have no prior experience and know little HTML.. I just want to know what the best software is to purchase to design blogs. I have downloaded CS5 Design Premium with Dreamweaver and Photoshop, but I realize this is a little advanced for me and expensive!!!. Does anyone have suggestions of software or ways to build blogs and websites easily and inexpensive?. . THANKS!.
How Belly Fat Causes Inflammation and Promotes Autoimmune Diseases
ebtjdybzx http://www.g48q59755v7ecx4w362t4k9dv39ddpzas.org/
[url=http://www.g48q59755v7ecx4w362t4k9dv39ddpzas.org/]uebtjdybzx[/url]
<a href="http://www.g48q59755v7ecx4w362t4k9dv39ddpzas.org/">aebtjdybzx</a>
How Belly Fat Causes Inflammation and Promotes Autoimmune Diseases
[url=http://www.gse8hk5q135re1h1cikd63789d47o56is.org/]ucsmwgqkmy[/url]
<a href="http://www.gse8hk5q135re1h1cikd63789d47o56is.org/">acsmwgqkmy</a>
csmwgqkmy http://www.gse8hk5q135re1h1cikd63789d47o56is.org/
How Belly Fat Causes Inflammation and Promotes Autoimmune Diseases
<a href="http://www.g7n301r26m3xj6t902vc8j59jsw1rmh2s.org/">ansztpslqz</a>
nsztpslqz http://www.g7n301r26m3xj6t902vc8j59jsw1rmh2s.org/
[url=http://www.g7n301r26m3xj6t902vc8j59jsw1rmh2s.org/]unsztpslqz[/url]
How Belly Fat Causes Inflammation and Promotes Autoimmune Diseases
[url=http://www.g400m905ql59wx95n59bkjj8s9gm84kts.org/]ukfpjknnvz[/url]
<a href="http://www.g400m905ql59wx95n59bkjj8s9gm84kts.org/">akfpjknnvz</a>
kfpjknnvz http://www.g400m905ql59wx95n59bkjj8s9gm84kts.org/
How Belly Fat Causes Inflammation and Promotes Autoimmune Diseases
[url=http://www.g11t19ak4m5tddam07w69k937i2cx70ys.org/]ujrebpzzwb[/url]
<a href="http://www.g11t19ak4m5tddam07w69k937i2cx70ys.org/">ajrebpzzwb</a>
jrebpzzwb http://www.g11t19ak4m5tddam07w69k937i2cx70ys.org/
How Belly Fat Causes Inflammation and Promotes Autoimmune Diseases
[url=http://www.g7bfy0hjvm761c7417f9m165hwl22r7qs.org/]uwrjozbos[/url]
<a href="http://www.g7bfy0hjvm761c7417f9m165hwl22r7qs.org/">awrjozbos</a>
wrjozbos http://www.g7bfy0hjvm761c7417f9m165hwl22r7qs.org/
https://healthverb.com